Understanding the Risks: Stem Cell Eye Treatment Controversies
Recent controversies highlight the significant risks of unproven stem cell eye treatments. This page explains a notable case and underscores why choosing FDA-approved clinical research and qualified specialists is crucial for your vision safety.
The Florida Case: A Warning About Stem Cell Eye Treatment Risks
In 2017, The New England Journal of Medicine reported that three women in Florida suffered permanent vision loss after receiving unproven stem cell injections for macular degeneration at a private clinic. This case exposes critical gaps in patient protection and the dangers of clinics operating outside rigorous clinical trials.
Key Takeaway: Stem cell eye treatment risks are real. Patients paid $5,000 each for a procedure that lacked FDA oversight and scientific validation, leading to blindness.
Patients Lose Sight After Stem Cells Are Injected Into Their Eyes
By DENISE GRADY
MARCH 15, 2017
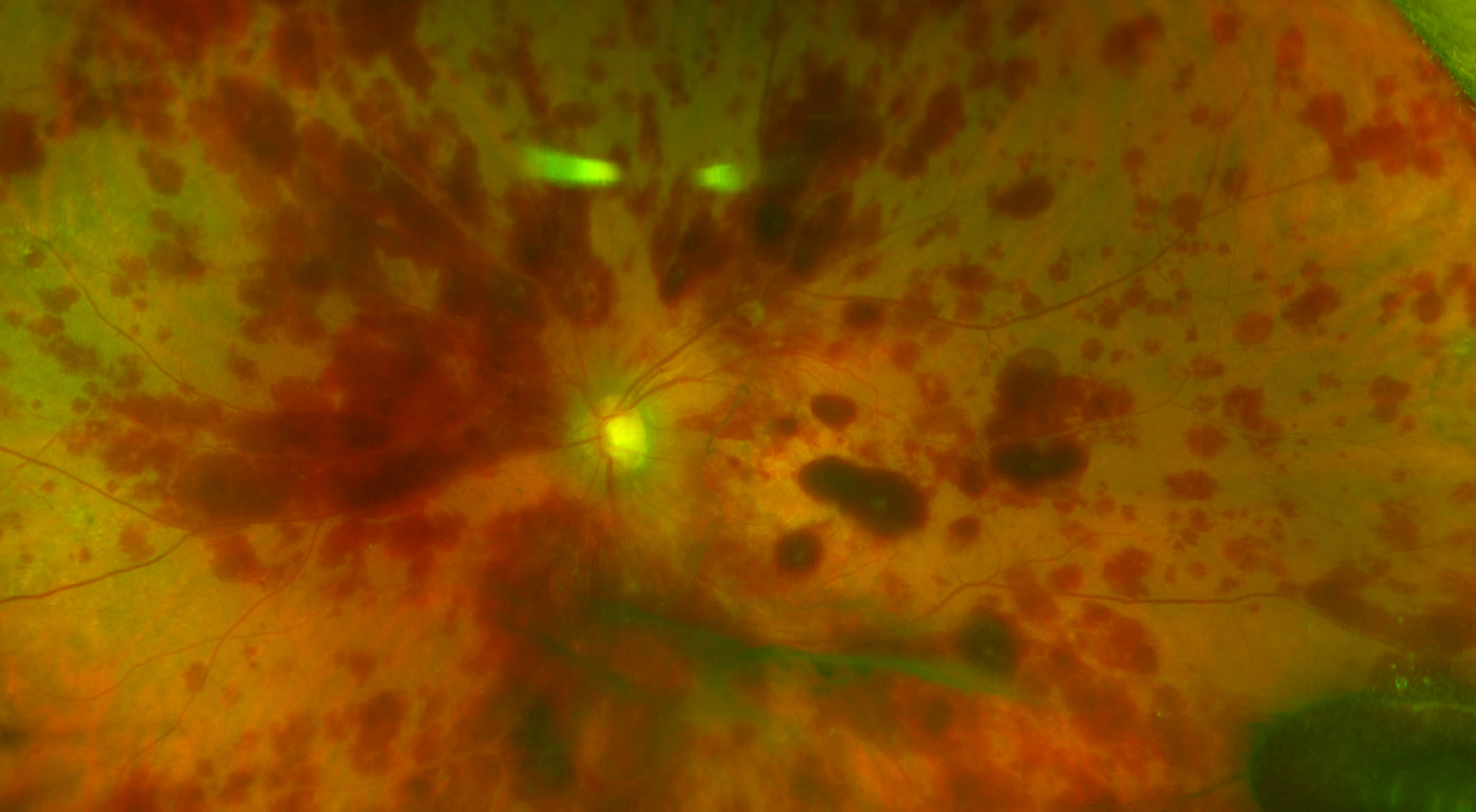
Credit: Dr. Thomas Albini
Three women suffered severe, permanent eye damage after stem cells were injected into their eyes, in an unproven treatment at a loosely regulated clinic in Florida, doctors reported in an article published Wednesday in The New England Journal of Medicine.
One, 72, went completely blind from the injections, and the others, 78 and 88, lost much of their eyesight. Before the procedure, all had some visual impairment but could see well enough to drive.
What Went Wrong?
- Procedure: The clinic used liposuction to extract stem cells from the patients’ fat, then injected them into both eyes simultaneously.
- Result: Patients experienced detached retinas and high eye pressure. One woman went completely blind.
- Regulation Gap: The clinic argued that using a patient’s own cells exempted it from FDA drug regulations.
All three women found U.S. Stem Cell because it had listed a study on a government website, clinicaltrials.gov — provided by the National Institutes of Health. Two later told doctors they thought they were participating in government-approved research. But no study ever took place, and the proposed study on the site had no government endorsement. Clinical trials do not need government approval to be listed on the website.
Legitimate research rarely, if ever, charges patients to participate, scientists say, so the fees should have been a red flag. But many people do not know that.

Credit: Scott McIntyre for The New York Times
Stem cells, which can develop into many different types of cells, are thought to have tremendous potential to repair or replace tissue damaged by disease, injury or aging. But so far, the F.D.A. has approved only a few stem-cell products to treat certain blood disorders.
Credit: Riccardo Cassiani-Ingoni/Science Source
Red Flags and How to Protect Yourself
This case reveals several warning signs for patients considering experimental stem cell therapies:
- Charging for Experimental Treatment: Legitimate clinical research rarely charges participants.
- Treating Both Eyes at Once: Ethical research treats one eye first to preserve vision if complications occur.
- Misleading Trial Listings: The clinic listed a “study” on a government website (ClinicalTrials.gov), which does not imply endorsement or approval.
Expert Insight from the FDA
In an accompanying article, FDA scientists warned that stem cells from fat “are being used in practice on the basis of minimal clinical evidence of safety or efficacy.”
The F.D.A. website warns “the hope that patients have for cures not yet available may leave them vulnerable to unscrupulous providers of stem-cell treatments that are illegal and potentially harmful.”
The F.D.A. article in The New England Journal of Medicine suggested that adverse events from stem-cell treatments “are probably much more common than is appreciated, because there is no reporting requirement when these therapies are administered outside clinical investigations.”
The Importance of FDA-Approved Research
While stem cell research for eye diseases holds great promise, progress must happen through controlled, ethical clinical trials. The FDA has approved only a few stem-cell products, primarily for blood disorders—not for intraocular use.
Choosing Safe, Ethical Eye Care
Your vision is irreplaceable. We strongly advise:
- Consulting a board-certified ophthalmologist before considering any experimental treatment.
- Verifying FDA approval status for any procedure or clinical trial.
- Being skeptical of “miracle cures” that bypass established medical channels.
Trustworthy Resources on Stem Cell Research
Read the Original NY Times Report: Patients Lose Sight After Stem Cells Are Injected Into Their Eyes
FDA Consumer Warning: FDA Warns About Stem Cell Therapies
Schedule a Consultation for Evidence-Based Care
If you or a loved one are exploring treatment options for macular degeneration or other eye conditions, our physicians can provide guidance on safe, effective, and scientifically validated pathways.